Search results
The page "Sodium-methoxide.net" does not exist. You can create a draft and submit it for review or request that a redirect be created, but consider checking the search results below to see whether the topic is already covered.
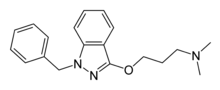
- an N-isocyanate #, which then cyclizes. Alkylation of the enol with sodium methoxide and 3-dimethylaminopropyl chloride gives benzydamine. Alternatively...13 KB (1,071 words) - 14:29, 24 November 2023
- reaction conditions are 80 °C and 40 atm. The most widely used base is sodium methoxide. Hydrolysis of the methyl formate produces formic acid: HCO2CH3 + H2O...38 KB (3,909 words) - 03:42, 22 May 2024
- converted to the corresponding alkene via its tosylhydrazone, using sodium methoxide (the Bamford–Stevens reaction) or an alkyllithium (the Shapiro reaction)...48 KB (5,110 words) - 13:29, 10 May 2024
- oxygen counts as a two-electron oxidation. Thus a net addition of H2O is a simple hydration with no net change in redox state and frequently occurs reversibly...19 KB (95 words) - 19:06, 28 April 2024
- Sodium hexafluoroacetylacetonate, NaC5HF6O2...38 KB (584 words) - 14:51, 14 May 2024
- chain fatty acids. The most common form uses methanol (converted to sodium methoxide) to produce methyl esters (commonly referred to as Fatty Acid Methyl...143 KB (15,730 words) - 06:14, 13 May 2024
- 2CO2 + 4H2O}}} Sodium + Methanol→Sodium methoxide + Hydrogen{\displaystyle {\text{Sodium + Methanol}}\rightarrow {\text{Sodium methoxide + Hydrogen}}}